You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
The ADAA has an obligation to disseminate knowledge in the field of dentistry. Sponsorship of a continuing education program by the ADAA does not necessarily imply endorsement of a particular philosophy, product, or technique.
Note: This course was adapted from an original course developed by Susan C. Dodd, RDH, BA.
Authors' Note: The term spit and smokeless tobacco will be used interchangeably throughout this course and refer to tobacco that is not burned but chewed or placed in the mouth. Smokeless tobacco users, usually, chew or suck (dip) the tobacco and spit out the tobacco juices that form. However, "spitless" tobacco has also been developed, and more recently smokeless cigarettes are being marketed.
Spit (smokeless) tobacco is a public health issue that gets limited attention. Unfortunately, there are segments of the Unites States population where use rates are high. While many health history questionnaires raise questions concerning smoking, many do not address others forms of tobacco use. Patients using this product should be identified, urged to quit and provided counseling and cessation interventions. Like cigarette smoking, chewing tobacco, snuff, or moist snuff produces an addiction to nicotine. This product is associated with both oral health and systemic health risks. Dental providers are in situation to show patients the damaging impact that this product has on oral health and assist patients in quitting.
Basics of Spit Tobacco
Types of Spit Tobacco
Spit tobacco, also known as smokeless tobacco, comes in two forms: chewing tobacco and snuff. The products differ with regard to the type and part of the tobacco plant used, method of curing and processing, moisture content, and additives. Snuff is often referred to as "dip" and, at present, is the most popular type of spit tobacco sold in the U.S. It is produced in two varieties, moist and dry, and is packaged in loose and packed forms. While snuff users in the U.S. prefer the moist type, dry snuff use can still be found in population groups such as the elderly and tobacco users outside of the United States.1
Like cigarettes, the ingredients in smokeless tobacco can cause many adverse health problems. Nicotine, the main ingredient, is highly addictive and has documented use as an insecticide. Manufacturers are able to control the amount of nicotine in their products by altering the pH level of the product. Various flavorings can also be added to the tobacco product. The amount of flavoring can be increased to entice younger users. Spit tobacco also contains varying amounts of sodium and sugar. These products alone place the user at an increased risk for health problems such as high blood pressure and dental disease.2
Chewing Tobacco
Chewing tobacco is sold in three forms: loose leaf, plugs and twists. Loose-leaf chewing tobacco, which is packaged in pouches, is produced from stripped and processed tobacco leaves that are stemmed and then cut or shredded into strips. Chewing tobacco can also be pressed into bricks and sold as plugs.1
A third form of chewing tobacco is produced from a dark tobacco leaf, cured, processed and twisted into strands or ropes and dried.1
Chewing tobacco is used by taking a portion of the product (called a "quid" or "chaw") putting it into the mouth, chewing long enough to soften the quid, then placing it between the cheek or lower lip and the gums. It is periodically chewed or sucked to extract the nicotine-laden juice. Users of chewing tobacco produce a large volume of tobacco juice or saliva, which must be periodically spit out or swallowed.3
Snuff
Dry snuff is produced from fire-cured tobacco. This tobacco is fermented after the curing process, allowed to dry and finely ground into a powder. With moisture content of less than 10%, dry snuff can be inhaled through the nose or placed in the mouth between the cheek and gum which requires spitting.1
Moist snuff is produced from tobacco that has been either air or fire cured, processed and finely ground or cut. The moisture content can be up to 50%, allowing the user to pinch a small amount of the tobacco or "dip" and place it between the lip or cheek and the gingiva. Moist snuff is sold loose in round cans or packaged in premeasured tea bag like packets.1 Each pinch or packet (depending on the brand) contains the same amount of nicotine as two to five cigarettes. It is the constant high level of nicotine in the blood that results in the intense addiction found in spit tobacco users.2
A newer form of smokeless tobacco has recently been introduced in the United States: dissolvables. These products are designed to dissolve slowly in the mouth and do not require spitting.1,4 These products do not look like conventional tobacco products, many even resemble candy which makes them marketable to younger populations.1
History of Spit Tobacco Use
It was not until the 15th century and the arrival of Europeans such as Christopher Columbus and Amerigo Vespucci that it was carried to the rest of the world. It was in Europe, especially among royalty and the privileged class, that dry snuff use became popular. It was inhaled by men and women alike who considered its use socially acceptable. By 1570, Europeans were growing a few varieties of tobacco and producing snuff that was eventually exported back to the American colonies.5
Tobacco was once so valuable that it could be used as currency. Early on in Europe, tobacco was believed to cure many different ailments. There were several books published on the subject. Tobacco was believed to cure anything from joint pain, epilepsy and even the plague. Chewing tobacco use was first documented in the 1700's. It was during this time that pipe smoking was also gaining popularity.5
In the early 1800s, spit tobacco was referred to as "The American Habit." It was common to see communal spittoons in public buildings and to see users spitting on the street and floors. Even the U.S. Congress had communal snuffboxes and cuspidors.5,6
In the early 1900's, spit tobacco use began to sharply decline and became socially unacceptable. This decline was precipitated by three events. One was the belief that tuberculosis could be spread by tobacco juice. The second was the subsequent law against public spitting. The third event was the invention of an efficient cigarette-rolling machine. This allowed for the mass production of inexpensive cigarettes.6
Spit tobacco remained a socially unacceptable practice until after the release of the 1964 Surgeon General's report that outlined the health dangers of cigarette smoking. The spit tobacco companies saw this as an opportunity to reintroduce their product as a safe alternative to cigarettes. Thus, began intensive mass media advertising using sports figures to show that their product was macho and a component of a successful athletic career. As a result of this very effective campaign, spit tobacco use regained its popularity and has been rising, especially among young people, ever since. In 2014 current smokeless tobacco use was highest in Wyoming, West Virginia, Montana, and Mississippi and lowest in Delaware, Hawaii, Massachusetts, California, Connecticut and Maryland.7
Advertising and Consumption
According to the Federal Trade Commissions (FTC) Report for the Year 2014, released in November 2016, the total amount of spit tobacco sold by manufacturers has decreased slightly from 128.04 million pounds in 2013 to 127.81 million pounds in 2014.8 Overall revenue from spit tobacco has risen 4.7%, increasing from $3.263 billion in 2013 to $3.416 in 20148 while expenditures on advertising increased to $6.8 million,8 a 16% rise from 2013 to 2014.8 Spending for magazine and outdoor advertising increased (change from raised), while dollars spent on direct mail decreased. The majority of advertising dollars, 72.4%, is spent on promotional allowances and price discounts with the hope of gaining new customers.8
In 2009, the Family Smoking Prevention and Tobacco Control Act permitted the Food and Drug Administration to regulate tobacco products and industry marketing practices. Currently, the Federal Food, Drug & Cosmetic Act (FD&C) has established a timeline for newly-regulated products to reach compliance which includes stopping the manufacture of products with modified risk claim by November 8, 2017 (unless there is a Modified Risk Tobacco Product order) and halt distribution by December 8, 2017.9 Currently, there are four required warning statements on labels of smokeless tobacco:9
WARNING: This product can cause mouth cancer.
WARNING: This product can cause gum disease and tooth loss.
WARNING: This product is not a safe alternative to cigarettes.
WARNING: Smokeless tobacco is addictive.
One manufacturer of snus requested that the FDA permit them to remove fist two required warnings and revise the third required warning to "substantially lower risks to health then cigarettes".10 This request to be designated as a Modified Risk Tobacco Product (MRTP) was denied.11 Had this request been successful, the manufacturers of snus would have the potential for a successful marketing campaign, enticing tobacco users away from cigarettes by promising lower health risks.
A documented strategy, developed by the largest manufacturer of spit tobacco in the United States, to graduate new, young users to stronger brands has been in place since 1984. The strategy has become known as the Graduation Method and has proven to be very successful. Designed to appeal to the very young potential user (10-12 yrs.), a product is marketed as pre-measured packets containing tobacco, sweeteners and flavoring.12 Designed for the beginner, the packets are sweeter and milder tasting, less likely to cause side effects such as nausea, and are easier to control in the mouth.12 After a period of time, the young user develops a tolerance to this lower nicotine product and "progresses" to the next level product that supplies more nicotine and a stronger tobacco flavor.12 Most habitual users will graduate to the strongest snuff with the highest levels of nicotine in order to satisfy their addiction.12 Once at this level, the user is addicted and would find quitting spit tobacco to be very difficult.
Other techniques used by the spit tobacco industry to attract and keep new customers include coupons and retail value-added promotions, public entertainment, sponsorship of sporting events and concerts, distribution of free samples, magazine advertising, and continuing to use the term "smokeless." Links to role models and athletes in the arenas of rodeo, car racing, baseball, football, soccer, fishing and hunting continue to be promoted by the spit tobacco industry in their appeal to new and younger potential users. There continues to be smokeless tobacco advertising directly marketed toward males, both young and old, insinuating that using smokeless tobacco is ‘manly' or typically enjoyed with other males. Additionally, highlighting the various flavors of smokeless tobacco products appeals to the younger population.13 Smokeless tobacco products are specifically marketed towards cigarette users as acceptable alternatives that may be used where smoking is banned and often carry the same brand name to further recognition.14
Over the past decade, the two largest manufacturers of cigarettes in the United States, R.J. Reynolds and Phillip Morris entered the spit tobacco market with a new generation of products. R.J. Reynolds is aggressively marketing a variation of snuff under the name of Camel® Snus in addition to other products designed to dissolve in the mouth. Phillip Morris has joined this market with Marlboro Snus and a marketing plan aimed at all tobacco users with the message that snus is a safer and cleaner than cigarettes. There was an increase of moist smokeless tobacco use with the release of snus products between the years 2006 - 2010.14
In 2008, R.J. Reynolds introduced three variations of dissolvable tobacco products, pellets, sticks and film strips, under the umbrella of Camel® Dissolvables. They contain between 60%-300% of nicotine found in cigarettes and are marketed to enable a tobacco user to maintain a tobacco habit in clean indoor air facilities and tobacco free zones. These products were formulated to resemble familiar and socially acceptable products such a candies, breath mints, breath freshening strips and flavored toothpicks.15
The dissolvable tobacco products were introduced and marketed in five U.S. cities.16 After five years, the sales success of the dissolvable products has been limited with surveys showing not only low sales numbers, but a lack of consumer awareness. In August, 2013, R. J. Reynolds announced that they were limiting the distribution and marketing of Camel® Dissolvables to two U.S. cities with no plans for expanding the markets.16 They also announced that they will continue to research and develop new smokeless products.16
Another marketing approach by tobacco companies is to market smokeless tobacco products that resemble cigarettes.17 Recently, Philip Morris has taken steps to become a leader in smoke free products by corporate restructuring and global marketing of both traditional and smoke free tobacco products.17 These actions, paired with the development of IQOS® (a tube that heats up sticks of tobacco instead of burning them), have at least one investment firm strongly recommending investing in Philip Morris.17 IQOS® is currently sold in 25 countries and has garnered a significant share of the tobacco market.18 In addition to the unknown long term health effects, a major concern is the marketability of IQOS®. The younger population could be attracted to the sleek design and new technology.18 Philip Morris is currently waiting on FDA approval in the United States.
Understanding the Dangers and Health Consequences of Smokeless Tobacco Use
Chemical Composition of Smokeless Tobacco
Processed tobacco contains over 2,500 chemical compounds, many of which have been directly linked to causing cancer in humans.19,20 More than 30 carcinogens exist in smokeless tobacco, including volatile and tobacco-specific nitrosamines, nitrosamino acids, polycyclic aromatic hydrocarbons, aldehydes, metals and polonium-210.21, 29 The oral use of smokeless tobacco is estimated to be the greatest external source of human exposure to nitrosamines which are causative factors in tobacco-related human cancers. Another chemical compound in tobacco is nicotine, which is an addictive drug.
Nicotine and Addiction
Nicotine is a plant product and naturally occurs in all tobacco plants. As in other forms of tobacco, smokeless tobacco delivers nicotine to the user's central nervous system. "It produces transient, dose-related effects dependent on the user's physiology. It acts to reinforce and control the user's behavior. Habitual users develop a tolerance to nicotine's effects and may increase the quantity of tobacco product used daily to reach their desired level.22 The pharmacological and behavior processes leading to nicotine dependence are like addiction to heroin and cocaine. As is the case for those other drugs of abuse, nicotine produces effects in the user that are considered desirable to the user.22
All drugs of abuse work on the pleasure center of the brain in the same way. The cells in those centers are changed in profound, long lasting ways. These drugs produce common effects on the production of dopamine, a neurotransmitter involved in experiencing pleasure. These effects include transient dose related changes in mood and feeling. Maintaining desired levels of these feelings is dependent upon continued administration of nicotine. As the level decreases, the user will often experience mood swings, irritability, impulsiveness, anger and depression. These withdrawal symptoms make cessation difficult in the first couple weeks of cessation efforts. The presence of nicotine in the body also causes skeletal muscle relaxation, cardiovascular effects and hormonal changes.23
Levels of nicotine vary depending on the form and brand of smokeless tobacco used. According to the CDC, tobacco users who dip or chew 8 to 10 times per day may be receiving the nicotine equivalent of 30 to 40 cigarettes per day. Each dip of snuff contains the same amount of nicotine as 2-5 cigarettes.24
Smokeless tobacco manufacturers regulate the amount of free nicotine in their product by adjusting the pH of the tobacco. The average pH of tobacco is 7.4, but adding alkaline additives, such as sodium bicarbonate, increases the ph. This increase will raise the amount and rate of absorption of free nicotine into the bloodstream. Studies done by the CDC, found the pH level in common smokeless tobacco brands varied from 5.24 in Hawken® Wintergreen to 8.35 in Kodiak® Wintergreen and Copenhagen® Snuff. These common brands have the highest levels of nicotine and are the most physiologically addictive. They also had the highest concentration of tobacco specific N-nitrosamines, at the time this study was conducted.25Tobacco companies manipulate the pH level of their smokeless tobacco products to increase the rate of absorption of nicotine from the oral mucosa into the bloodstream. Therefore, their product attribute to a more profound increase in mood and pleasure and become more desirable and addictive.
Smokeless tobacco users develop a nicotine addiction like that of cigarette smokers and have been found to absorb at least as much nicotine as smokers. The rate of absorption from snuff is especially rapid as the high pH of saliva provides a favorable environment for absorption through the oral mucosal.26 Young users appear to rapidly develop a physical dependence from smokeless tobacco use and experience withdrawal symptoms after a relatively short time, as a regular user. The more addicted an adolescent is, the more difficult it will be for them to quit and the more likely they are to become adult users. Success rates in smokeless tobacco cessation attempts by addicted users remain low. In fact, regardless of age, 74% of daily users reported that one of the reasons they continued to use it was because it was hard to quit. The American Cancer Society reports that quitting smokeless tobacco is as difficult as smoking cessation.27
Cardiovascular, Endocrine and Gastric Health Concerns
The nicotine in smokeless tobacco is a poisonous chemical which can cause serious health consequences. In addition to its addictive quality, it has been found to activate the sympathetic nervous system resulting in constriction of the blood vessels, reduction of coronary blood flow and an increased risk of vascular thrombosis. The effects on the cardiovascular system can lead to an increased heart rate (10-20bpm), increased blood pressure (5-10mmHg), and reduced arterial blood flow causing dizziness and decreased endurance and stamina. An increase in blood clotting factors may lead to the formation of clots in already constricted vessels, causing a stroke or heart attack.28
Studies have shown nicotine to affect the hormonal and metabolic systems. It increases blood glucose levels leading to higher insulin levels and consequential energy swings. The level of the hormone cortisol, involved in wound healing and swelling, is increased by nicotine. This abnormal rise disrupts the normal healing process. Digestive problems have been linked to the nicotine and other chemicals found in tobacco juice. While smokeless tobacco users attempt to spit out the accumulating tobacco juice and saliva, over 50% of the juice is swallowed. The toxins in the juice cause problems such as acid reflux, esophageal ulcers, peptic ulcers and a potential risk for cancer of the larynx, esophagus, and stomach.28
Cancer
Research regarding smokeless tobacco and cancer report that use of smokeless tobacco increases risk for oral, pancreatic and esophageal cancer. A summary of research by Bofetta, Petal regarding cancer risk from smokeless tobacco use states "Although epidemiological data from the USA and Asia show a raised risk of oral cancer in those who use smokeless tobacco (overall relative risk 2•6 [95% CI 1•3-5•2]), these are not confirmed in northern European studies (1•0 [0•7-1•3]). Increased risks of esophageal cancer (1•6 [1•1-2•3]) and pancreatic cancer (1•6 [1•1-2•2]) are shown with smokeless tobacco use in northern European studies. In India and Sudan, more than 50% of oral cancers are attributed to smokeless tobacco products used in those countries, as are about 4% of oral cancers in US men and 20% of esophageal and pancreatic cancers in Swedish men."29
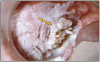
As a dental health provider, it is important to include an oral cancer assessment on all patients, especially those who use tobacco products. The most common sites for oral cancerous lesions to develop are the lateral border of the tongue (20%), gingiva and cheek (18%), floor of the mouth (12%), lips (11%), and the salivary glands (8%). Cancer associated with smokeless tobacco is often located where the quid or product is held, on the buccal and alveolar surfaces. The two types of cancer associated with smokeless tobacco use are verrucous and squamous cell carcinoma; however, the prevalence is low.30,34
Squamous cell carcinoma is a malignant neoplasm that exhibits squamous differentiation characterized by the formation of keratin or the presence of intercellular bridges.30 Intraoral lesions are white and wrinkled in appearance with intermittent red lesions.
Verrucous carcinoma is a warty variant of squamous cell carcinoma characterized by a predominantly outward overgrowth of well-differentiated keratinized epithelium.30 Intraoral lesions appear keratinized with white finger-like projections.
There are several risk factors associated with developing oral and pharyngeal cancer. According to the October 2008 report from the National Institute of Dental and Craniofacial Research (NIDCR), most cases of oral cancer are linked to tobacco, heavy alcohol use or the use of both tobacco and alcohol together. There are a growing number of cases being diagnosed in people under 50 years old without the historic risk factors of tobacco and alcohol use. Infection with the sexually transmitted human papilloma virus (HPV 16) has been linked to this subset of oral cancers. The risk for cancer of the lips increases with prolonged exposure to ultraviolet radiation. The carcinogenic agents penetrate and damage the DNA in the cells, causing the DNA to malfunction and accumulate abnormal cells resulting in the formation of a cancerous tumor. As the damage increases, the cells will metastasize.31
Oral Effects of Smokeless Tobacco
The most notable health consequences associated with smokeless tobacco use are those seen in the oral cavity. These effects can be recognized by the patient as well as the clinician and provide excellent opportunities to educate the user in the dangers of the tobacco use.
Leukoplakia/Keratosis
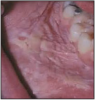
Leukoplakia, also known as a localized epithelial abnormality, can appear after only a few months of regular smokeless tobacco use. The lesions take several forms, requiring a biopsy for accurate diagnosis. Characteristics include a white color change that cannot be wiped off with gauze, granularity of the surface, wrinkling, corrugation and/or a thickening of the mucous membrane. There may also be red areas intermingled with the white areas and it may be modular in appearance.32 The frequency and duration of smokeless tobacco use are positively related to the prevalence of leukoplakia. In heavy, habitual users, over 50% develop oral mucosal abnormalities within 3.3 years of use. The frequency of use (more than 15 minutes at a time for several times a day) and overall length of use are important factors in the likelihood of developing lesions.27 Leukoplakia lesions are prevalent with smokeless tobacco users in India and South Asia.29
Tobacco-associated keratosis is an ill-defined area of white thickening at the sites at which oral smokeless tobacco is habitually placed; most commonly, these areas involve the mandibular labial and buccal mucosal folds. The continued use of smokeless tobacco causes the affected areas to become corrugated and grey in color. Microscopically, smokeless tobacco keratosis show hyperkeratosis and acanthosis of the mucosal epithelium. True epithelial dysplasia is uncommon; when dysplasia is found, it is usually mild in degree. Such lesions typically occur in the buccal or labial vestibule where the tobacco is held, but they can also extend onto the adjacent gingiva and buccal mucosa.30
Most white lesions will dissipate within 6 weeks after discontinuing tobacco use. Up to 75% will regress if the tobacco is removed from the site. However, it is vital that a patient with a white lesion is re-examined in 2 weeks to be sure that healing is occurring, and the tobacco has been removed from the site. If there is no change, a biopsy should be performed immediately. The patient who continues to use tobacco should be taught to do a self-oral cancer screening and to return to his health care professional for regular exams.33 Not all lesions are discovered before they undergo transformation to a dysplastic state. According to the American Cancer Society (ACS), 3-5% of the leukoplakia are cancerous or exhibit changes that progress to cancer within 10 years if not properly treated.27
Halitosis, Teeth Staining & Abrasion, Gingival Attachment Loss, Dental Caries
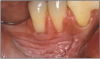
Smokeless tobacco use can result in halitosis, staining, abrasion of the teeth, gum recession, and dental caries.27 Smokeless tobacco manufacturers add other compounds to their products to achieve the flavors their customers want, such as cane sugar, honey, licorice, or rum and abrasives. These components of smokeless tobacco, along with poor oral hygiene can cause extensive gingival attachment loss and carious lesions on the exposed root surface. Focal gingival recession with periodontal attachment loss and bone erosion occurs frequently on the facial surfaces of the teeth where the tobacco is held. This recession is a result of the irritation caused by the toxic chemicals and abrasives found in smokeless tobacco. It is not uncommon to find substantial amounts of root surfaces exposed at the tobacco placement site. With the root surface exposed, there is an increased risk of hot and cold sensitivity.28 Bleeding is less common because of the vasoconstriction properties of nicotine.27
Additional characteristics of long-term smokeless tobacco use are stained teeth and halitosis. The tobacco and the juice produced can penetrate the tooth enamel and restorative material, resulting in a brown or yellow darkening of the teeth. These stains, in the presence of poor oral hygiene, can also result in a dark brown line along the gingival margins of the affected teeth.28 Halitosis is the most commonly recognized negative consequence of smokeless tobacco use.28 It is a result of the constant presence of tobacco juice in the user's mouth. In addition, the need to continually spit out excess juice and saliva is offensive and unhygienic. Smokeless tobacco users often find pieces of tobacco lodged between their teeth, in the buccal folds and on the lips.
The abrasives found in smokeless tobacco can also wear away portions of the occlusal/incisal surfaces of the teeth resulting in a weakening and eventual chipping of those surfaces. The abrasives also irritate the gingival tissue, allowing for more efficient absorption of the carcinogenic agents and nicotine found in the tobacco. Because of the chemicals and sodium in smokeless tobacco, users suffer a lessening of their senses of taste and smell. This can lead to unhealthy eating habits. Excess sodium may augment the effect of nicotine on elevating blood pressure.28
According to a study conducted by Dr. Scott Tomar and Dr. Deborah Winn for the National Institute of Dental Research (NIDR), individuals who use chewing tobacco have significantly more decay on the crowns and roots of their teeth than nonusers. The study showed that men (over 18 years) who use chewing tobacco exclusively were four times more likely than a nonuser to have one or more decayed or filled root surfaces. The more packages chewed, the greater the chance of having decayed or filled surfaces. This increased risk is associated with the high sugar content of the tobacco and the way it is used. Typically, a wad of chew is held in the mouth for 30 minutes at a time, and then replaced with a new wad, thus exposing the teeth to the tobacco and sugar for many hours a day.35 Therefore, smokeless tobacco using patients need counseling on its cariogenic effects to prevent continued damage to tooth structure, oral tissues and overall health. Due to these substantial health and oral cavity risks with use of smokeless tobacco, dental providers need to assess each patient's tobacco use on a regular basis and encourage/assist with the patient's cessation from all tobacco products.
The Dental Professional's Role in Spit Tobacco Prevention
Regrettably, there are many geographical areas in the United States where the spit tobacco use rate is much higher than the 3.45% national use rate (6.7% men and 0.3% women) recorded in 2014. In 2015, the overall high school male use rate was 10.0%.36 While many states have a much higher use rate than this national average, many states will also will not meet the 2020 Healthy Goal of reducing spit tobacco rates to 0.3%.37
Dental health care workers can be a unique source of information and guidance to aid in efforts to reduce spit tobacco use in either the community or private practice setting. A review of 14 studies which included over 10,500 participants suggests that tobacco intervention by dental personnel increased abstinence rates among both cigarette and smokeless (spit) tobacco users. While differences between the studies restricted the ability to make conclusive recommendations regarding the interventions that should be included in practice or community settings, behavioral counselling and an oral examination was a consistent intervention among the studies.38 Prior research suggests that personalizing the message to the patient regarding the impact tobacco is having on his/her mouth is an important stimulus in initiating cessation attempts.39
A 2008 U.S. Public Health Report called "A Clinical Practice Guideline for Treating Tobacco Use and Dependence" identified ten tobacco treatment guidelines. Dental professionals to be cognizant of several of these guidelines, particularly:40
(1) Tobacco dependence is a chronic disease that often requires repeated intervention and multiple attempts to quit.
(2) Tobacco dependence treatments are effective across a broad range of populations.
(3) It is essential that clinicians and healthcare delivery systems consistently identify and document tobacco-use status and treat every tobacco user seen in a healthcare setting.
(4) Brief tobacco-dependence treatment is effective.
(5) Telephone quitline counseling is effective with diverse populations and has broad reach. Therefore, clinicians and healthcare delivery systems should both ensure patient access to quitlines and promote quitline use.
(6) Individual, group, and telephone counseling are effective and their effectiveness increases with treatment intensity. Two components of counseling are especially effective and clinicians should use these when counseling patients making a quit attempt:
• Practical counseling (problem-solving/skills training)
• Social support delivered as part of treatment
Dental providers should actively participate in tobacco cessation strategies both at the community level and in private practices. The protocol for addressing this issue must be consistent and strong. There is no safe level of tobacco use!
Individuals that use tobacco should be advised to stop and measures should be in place to assist those interested with cessation. Personalizing messages to the patient regarding the impact that tobacco is having on their mouth can be a powerful motivator in helping patients take the first step towards cessation.
Tobacco is the number one cause of preventable death and disease in the United States.41 Dental providers see the impact of this product in patients' mouths every day and the community and patients need their advice and information to help keep them healthy. The Centers for Disease and Control Prevention has provided the below information to help dental personnel to help patients become tobacco free. While some items relate to smoking there is also important information provided at the links regarding available brochures, fact sheets, and quitlines for spit tobacco.42
https://www.cdc.gov/tobacco/campaign/tips/partners/health/dental/index.html
https://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/use_us/index.htm
Other resources include the American Cancer Society:43
https://www.cancer.org/healthy/stay-away-from-tobacco/guide-quitting-smoking/deciding-to-quit-smokeless-tobacco-and-making-a-plan.html
The National Spit Tobacco Education Program:44
https://oralhealthamerica.org/our-work/nstep/
The National Cancer Institute:45
https://www.cancer.gov/about-cancer/causes-prevention/risk/tobacco/smokeless-fact-sheet
Chewing Tobacco: Not a safe product-Mayo Clinic46
https://www.mayoclinic.org/healthy-lifestyle/quit-smoking/in-depth/chewing-tobacco/art-20047428
Glossary
Abrasion - wearing away of the surfaces of the teeth by friction; excessive wear can be caused by the sand or grit present in spit tobacco.
Addiction - compulsive use of a habit-forming drug or substance accompanied by increasing frequency and intensity in order to experience the desired effects.
Carcinogenic - anything that causes cancer.
Chewing tobacco - tobacco sold as loose-leaf, plugs, bricks, and twists; held in the mouth between the cheek and gums and is chewed or sucked to extract the nicotine.
Dependence, physiological - condition in which the body relies upon a drug or substance, resulting in specific body cell alterations and in a state where continued use is necessary to maintain the body's state of normalcy and balance.
Dependence, psychological - condition in which the user of a drug considers the effects it produces essential to maintain an optimal state of wellbeing.
Dipping - act of placing moist snuff in the mouth; dip is considered to be the amount of tobacco held between the thumb and forefinger.
Dissolvable tobacco products - tobacco products that dissolve in the mouth and do not require spitting or discarding of the product.
Dopamine - a monoamine neurotransmitter that is formed during the synthesis of norepinephrine and is essential to the normal functioning of the central nervous system.
Drug - substance that can alter mood, perception or consciousness.
Gingivitis - inflammation of the gingiva tissue resulting in redness, swelling, bleeding, and often discomfort.
Graduation method - tobacco users start with milder tasting and flavored products and eventually graduate to less flavored items that often contain more nicotine, resulting in a life-long addiction.
Halitosis - unpleasant breath.
Human papilloma virus - a virus with more than100 subtypes that cause diseases in humans ranging from common warts to cervical cancer.
Insecticide - a substance used for killing insects.
IQOS®™ - smokeless cigarette developed by Philip Morris.
Keratosis - a horny growth, especially on the skin.
Leukoplakia - formation of white, irregularly shaped, thickened patches in the oral cavity; found at the site of spit tobacco placement; may become squamous cell carcinomas (3%-5% of the cases).
Nicotine - an alkaloid derived from tobacco that acts as the addictive agent in both smoking tobacco and spit tobacco.
N-nitrosamines - cancer-causing chemicals found in cured tobacco products.
Oral cancer - cancer of the oral cavity, including the lips, tongue, gingival tissue, hard palate, soft palate, floor of the mouth, roof of the mouth, mucosal membrane, and oropharynx.
PH level - degree of alkalinity or acidity of a solution; values above 7 indicate alkalinity and result in a more efficient absorption of free nicotine found in spit tobacco; pH level of tobacco can be regulated by the manufacturer with the use of alkaline additives, such as sodium bicarbonate.
Recession - exposure in the roots of the teeth caused by a loss of gingival tissue.
Squamous carcinoma - is a type of skin cancer that begins in the squamous cells. Squamous cells are the thin, flat cells that make up the epidermis, or the outermost layer of the skin.
Smokeless tobacco - tobacco that is not smoked, also known as spit tobacco; term used by the tobacco industry to promote its deadly product as one that is safer than cigarettes.
Snuff, dry - dry, powdery tobacco product that is usually sniffed into the nose.
Snuff, moist - finely cut and ground moist tobacco product that is placed in the mouth between the teeth and gingiva or lip. It is also known as "dip".
Snus- smokeless or spitless tobacco that is usually found in patches.
Surgeon General - chief medical officer of the United States Army, Air Force, or Public Health Service.
Tolerance - capacity to assimilate a drug, such as nicotine, continuously or in increasing larger doses to achieve a constant effect.
Twist- dried tobacco leaves that are twisted into ropes and sold in pouches.
Withdrawal syndrome - intense physiological disturbances that occur when abruptly discontinuing the administration of a drug that, by its prolonged use, induced physical dependence; signs and symptoms include nausea, irritability, insomnia, restlessness, and headache.
Verrucous carcinoma - is an uncommon variant of squamous cell carcinoma. This form of cancer is often seen in those who chew tobacco or use snuff orally, so much so that it is sometimes referred to as "Snuff dipper's cancer."
References
1. Centers for Disease Control & Prevention; Smokeless Tobacco: Products & Marketing. Dec 2016. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/products_marketing/index.htm. Accessed: 10-29-2017
2.National Spit Tobacco Education Program. What You Need to Know: Spit Tobacco Facts. 2017. http://oralhealthamerica.org/wp-content/uploads/What-you-need-to-know.pdf. Accessed 10-29-2017
3. Mayo Clinic; Chewing Tobacco: Not a safe product. Sept 2017. https://www.mayoclinic.org/healthy-lifestyle/quit-smoking/in-depth/chewing-tobacco/art-20047428. Accessed 10-29-2017
4. United States Food and Drug Administration; Dissolvable Tobacco Products. June 2017. https://www.fda.gov/TobaccoProducts/Labeling/ProductsIngredientsComponents/ucm482569.htm. Accessed: 11-3-2017
5. National Park Service; Tobacco: The Early History of a New World Crop. Feb 2015. https://www.nps.gov/jame/learn/historyculture/tobacco-the-early-history-of-a-new-world-crop.htm Accessed 10-29-2017
6. tobaccofreeutah.org; History of Smokeless Tobacco. Dec 2007. http://tobaccofreeutah.org/pdfs/hissmkls.pdf. Accessed 10-29-2017
7. Centers for Disease Control and Prevention. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/use_us/index.htm. Accessed 10-25-17
8. Federal Trade Commission Cigarette Report for 2014 and Federal Trade Commission Smokeless Tobacco Report for 2014. (2016, November 16). https://www.ftc.gov/reports/federal-trade-commission-cigarette-report-2014-federal-trade-commission-smokeless-tobacco. Accessed 10-25-17
9. US Food & Drug Administration Tobacco Products: Advertising & Promotion. (2017, October https://www.fda.gov/TobaccoProducts/Labeling/MarketingandAdvertising/default.htm. Accessed 10-30-17
10. FDA News Release: FDA Takes Action on Applications Seeking to Market Modified Risk Tobacco Products. (2016, December 14). https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm533219.htm. Accessed 10-30-17
11. FDA Denies Modified Risk Tobacco Product Status for Snus. (n.d.). Retrieved October 30, 2017, from https://www.acsh.org/news/2016/12/14/fda-denies-modified-risk-tobacco-product-status-snus-10592
12. Connolly, G. N. (1995). The marketing of nicotine addiction by one oral snuff manufacturer. Tobacco Control, 4(1), 73-79. doi:10.1136/tc.4.1.73
13. Delnevo, C. D., Wackowski, O. A., Giovenco, D. P., Manderski, M. T., Hrywna, M., & Ling, P. M. (2012). Examining market trends in the United States smokeless tobacco use: 2005-2011. Tobacco Control, 23(2), 107-112. doi:10.1136/tobaccocontrol-2012-050739
14. The Health Consequences of Smoking-50 Years of Progress: A Report of the Surgeon General, 2014. (n.d.). https://www.surgeongeneral.gov/library/reports/50-years-of-progress/index.html. Accessed 10-30-17
15. Southwell, B. G., Kim, A. E., Tessman, G. K., Macmonegle, A. J., Choiniere, C. J., Evans, S. E., & Johnson, R. D. (2012). The Marketing of Dissolvable Tobacco: Social Science and Public Policy Research Needs. American Journal of Health Promotion, 26(6), 331-332. doi:10.4278/ajhp.111004-cit-357
16. Craver, R. (2013, July 31). R.J. Reynolds scales back marketing of dissolvable tobacco products. Winston-Salem Journal http://www.journalnow.com/business/business_news/local/r-j-reynolds-scales-back-marketing-of-dissolvable-tobacco- products/article_9d001b58-f9f2-11e2-8fad-0019bb30f31a.html. Accessed 10-30-17
17. LaVito, A. (2017, September 28). Philip Morris shuffles management on its quest toward a smoke-free future. From https://www.cnbc.com/2017/09/28/philip-morris-shuffles-management-on-its-quest-toward-a-smoke-free-future.html. Accessed 10-30-17
18. Wan, W. (2017, August 14). Big tobacco's new cigarette is sleek, smokeless - but is it actually healthier? http://www.chicagotribune.com/business/ct-smokeless-cigarette-IQOS®™-20170814-story.html Accessed 10-30-17
19. National Cancer Institute (NCI), "Smokeless Tobacco and Cancer," Accessed September 9, 2014. http://www.cancer.gov/about-cancer/causes-prevention/risk/tobacco/smokeless-fact-sheet#r1. See also: NIH, NCI, Smoking and Tobacco Control Monograph 2: Smokeless Tobacco or Health: An International Perspective, September 1992, http://cancercontrol.cancer.gov/tcrb/monographs/2/m2_complete.pdf.
20. National Toxicology Program, Public Health Service, HHS, Report on Carcinogens, Thirteenth Edition, October 2014, http://ntp.niehs.nih.gov/ntp/roc/content/profiles/tobaccorelatedexposures.pdf
21. National Toxicology Program, Public Health Service, HHS, Report on Carcinogens, Thirteenth Edition, October 2014, http://ntp.niehs.nih.gov/ntp/roc/content/profiles/tobaccorelatedexposures.pdf
22. Hatsukami DK, Stead LF, Gupta PC. Tobacco Addiction. Lancet 2008; 371(9629):2027-2038
23. NCI and U.S. Centers for Disease Control and Prevention (CDC), Smokeless Tobacco and Public Health: A Global Perspective, Bethesda, MD: HHS, CDC, National Institutes of Health (NIH), NCI, NIH Publication No. 14-7983, 2014. National Cancer Institute, "Smokeless Tobacco and Cancer," Accessed September 9, 2014, http://www.cancer.gov/about-cancer/causes-prevention/risk/tobacco/smokeless- fact-sheet#r1. See also: International Agency for Research on Cancer. A Review of Human Carcinogens: Personal Habits and Indoor Combustions. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 100E (2012), http://monographs.iarc.fr/ENG/Monographs/vol100E/mono100E-8.pdf.
24. Klein, J.A., Christen, A.G., Christen, J.A., McDonald, J.L., Guba, C.J. "Understanding nicotine addiction and intervention techniques for the dental professional." ADAA Continuing Education Course: Chicago: 1990.
25. MMWR: Determination of Nicotine, pH, and Moisture Content of Six U.S. Commercial Moist Snuff Products -- Florida, January-February 1999 Vol 48. No 19
26. Richter,P, Spierto,F. "Surveillance of smokeless tobacco nicotine, pH, moisture, and unprotonated nicotine content" Nicotine & Tobacco Research Volume 5, Number 6 (December 2003).
27. https://www.cancer.org/cancer/cancer-causes/tobacco-and-cancer/why-people-start-using-tobacco.html
28. The Health Consequences of Using Smokeless Tobacco: A Report of the Advisory Committee to the Surgeon General." U.S. Dept. of Health and Human Services. 1986
29. Boffetta P, Hecht S, Gray N, Gupta P, Straif K: Smokeless tobacco and cancer. Lancet Oncol. 2008, 9: 667-675. 10.1016/S1470-2045(08)70173-6.
30. Gupta J et al, Smokeless Tobacco and Oral Cancer: A review. Indian Journal of Science. Vol 3 July 2012
31. "Oral Cancer Causes and Symptoms & The Oral Cancer Exam." National Institute of Dental and Craniofacial Research National Oral Health Information Clearinghouse." NIH Publication, October 2008.
32. Oral lesions from smokeless tobacco dissipate after stopping use of tobacco products, study concludes." JADA, July. 1999.
33. Gupta, P. C., An epidemiologic assessment of cancer risk in oral precancerous lesions in India with special reference to nodular leukoplakia. Cancer 1989 June 1; 63(11); 2247-52
34. Kaugars GE, Brandt RB, Chan W, Carcaise-Edinboro P. Evaluation of risk factors in smokeless tobacco associated oral lesions. Oral Surg Oral Med Oral Pathol 1991; 72:326-31
35. Tomar, S and Winnn, D. "Chewing tobacco Use and Dental Caries Among US men" JADA, Vol 130, Issue 11; 1999
36. Office on Smoking and Health, National Center for Chronic Disease Prevention and Health Promotion; https://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/use_us/. Accessed 10-28-17
37. Office of Disease Prevention and Health Promotion; https://www.healthypeople.gov/2020/topics-objectives/topic/tobacco-use/objectives. Accessed 10-28-17
38. Carr AB, Ebbert J. Interventions for tobacco cessation in the dental setting. Cochrane Database of Systematic Reviews 2012, Issue 6. Art. No.: CD005084. DOI: 10.1002/14651858. CD005084.pub3; http://www.cochrane.org/CD005084/TOBACCO_can-interventions-delivered-by-dental-professionals-help-tobacco-users-to-quit. Accessed 10-29-17
39. J Dent Educ 2001 Apr; 65 (4) 354-63. Tobacco cessation through dental office settings, Gordon JS, Severson, HH; https://www.ncbi.nlm.nih.gov/pubmed/11336121(3). Accessed 10-28-17
40. Am J Prev Med 2008 Aug: 35(2):158-176; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4465757/. Accessed 10-28-17
41. Center for Disease Control and Prevention; https://www.cdc.gov/tobacco/campaign/tips/partners/health/dental/index.html. Accessed 10-29-17
42. Center for Disease Control and Prevention; https://www.cdc.gov/tobacco/data_statistics/fact_sheets/smokeless/use_us/index.htm. Accessed 10-29-17
43. The American Cancer Society; https://www.cancer.org/healthy/stay-away-from-tobacco/guide-quitting-smoking/deciding-to-quit-smokeless-tobacco-and-making-a-plan.html. Accessed 10-29-17
44. Oral Health America, National Spit Tobacco Education Project; https://oralhealthamerica.org/our-work/nstep/. Accessed 10/29/17
45. National Cancer Institute of the National Institutes of Health; https://www.cancer.gov/about-cancer/causes-prevention/risk/tobacco/smokeless-fact-sheet Accessed10-29-17
46. Mayo Clinic Healthy Lifestyle; https://www.mayoclinic.org/healthy-lifestyle/quit-smoking/in-depth/chewing-tobacco/art-20047428. Accessed 10-29-17
About the Authors
Susan Morgan, DDS, MAGD, CTTS
Susan Morgan received her dental hygiene and dental degree from West Virginia University. For approximately 10 years she treated low-income children, many of whom were already addicted to tobacco products. In 2000, as a faculty member in the Department of Periodontics, she implemented a tobacco cessation program in the WVU School of Dentistry. She has trained over 700 dental students to provide tobacco treatment strategies, and she has published the initial results of the progress of this program in the Journal of Dental Education. In 2010 she received her Tobacco Treatment Certification at the Mayo Clinic. She is currently a professor in the WVU School of Dentistry, and she has presented courses on tobacco cessation both locally and nationally.
M. Suann Gaydos, RDH, MSDH, CTTS
M. Suann Gaydos is a master's level trained dental hygienist and Tobacco Treatment Specialist who teaches tobacco treatment strategies to future health care providers. Suann was trained as a Tobacco Treatment Specialist in 2010 and has been involved in presenting tobacco treatment continuing education programs both locally and nationally. As a dental hygiene clinical supervisor, she provides students with the skills necessary to provide effective tobacco cessation counseling and support. She is currently a dental hygiene faculty member at West Virginia University's School of Dentistry.
Amy Funk, RDH, MSDH, TTS
Amy Funk is a master's level trained dental hygienist. Amy was trained as a Tobacco Treatment Specialist in 2010 and has been involved in presenting tobacco treatment continuing education programs both locally and nationally. She also directs the Dental Hygiene program at WVU School of Dentistry.
Ashlee Sowards, RDH, BSDH, MSDH, TTS
Ashlee Sowards received her master's in Dental Hygiene from West Virginia University, with her master's thesis title being, "Oral Cancer Screening Techniques Utilized by West Virginia Dental Hygienists." In April 2013 she completed her Tobacco Treatment Specialist training at the University of New Jersey School of Medicine and Dentistry. As a dental hygiene clinical supervisor, she provides students with the skills necessary to talk with their patients about tobacco dependence and offer cessation support. She is currently on the dental hygiene faculty at WVU School of Dentistry.