You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Introduction
Instruments are a major asset and represent a significant share of the total capital investment of a dental practice. It is important to understand the importance of ensuring proper processing and maintenance to retain the instrument value for many years. During daily use, and over time, dental instruments are subject to surface changes due to chemical, thermal, and/or physical impacts. It is important to stress that following manufacturer's instructions on instrument care is critical, and the manufacturer will always know the composition of instruments it manufactures. The dental assistant who understands the material and its characteristics, and who knows how to handle the product will be able to extend the life of every instrument.
INSTRUMENT COMPOSITION
Stainless Steel
Contrary to widely held views, stainless steel is not indestructible and is susceptible to a wide range of potential attacks by mechanical, thermal, or chemical agents. Stainless steel instruments contain various percentages of carbon and chromium, a steel alloy that is highly resistant to stains, corrosion, and rust. Probes, mirrors, explorers, extraction forceps, and orthodontic pliers are among the dental instruments usually made from stainless steel. These instruments will discolor and corrode when exposed to higher than recommended chemical concentrations, certain chemicals, or higher than recommended heat. Stainless steel is extremely damaged by chlorides (bleach) when used in the cleaning process and will quickly deteriorate.
Chromium and Nickel
Steel alloys with a higher percentage of chromium are more resistant to scratches, while alloys with a higher percentage of nickel have a smoother, more polished appearance. Corrosion resistance of stainless steel is enhanced by increased chromium content and the addition of other elements such as molybdenum, nickel, and nitrogen. Chromium and nickel are common components in steel alloys, but are primarily restricted to dental instruments not used for the actual cutting of tooth structures. Stainless steel orthodontic materials and stainless steel crowns (SSC) are the two major devices in pediatric dentistry that contain nickel.
Carbon Steel
Carbon steel is harder than stainless steel but is more susceptible to corrosion. Many dental instruments with sharp‐cutting edges, such as scalpels, are made of carbon steel because an alloy with a higher percentage of carbon will make a tool that is both harder and better able to maintain a sharp edge for a longer period of time.
Tungsten Carbide
Tungsten carbide is used to make burs that fit into dental handpieces. It is a good choice for burs because it is an extremely durable metal, and it is able to withstand the high heat that is generated by the rotation of the drill.
Other Instrument Composition
Implant instruments can be made of titanium, gold alloy, brass, plastic, stainless steel, and carbide steel. Specific manufacturer instructions should be followed explicitly when precleaning, cleaning, processing, and storing these instruments.
Composite instruments, whether stainless steel or XTS, should be protected to preserve the non‐stick surface by always wiping off any composite material from the working end with a 2x2 alcohol gauze (even if the substance is not evident) to promote straightforward cleaning and avoid permanent impairment of the exterior finish. Remember, new generation composite materials cure very rapidly, even with the operatory light.
Anodized aluminum Goldstein and Felt instruments cannot be ultrasonically cleaned, nor should they ever be placed in alkaline or iodophor solutions. They should be cleaned by hand or automated washer, and wiped off of any composite material while it is still moist (within 5 minutes).These instruments should be separated from the set‐up before the cleaning process begins because they have a special non‐stick surface that can and will be permanently harmed if scratched by other instruments or come in contact with cleaning or disinfecting solutions. If the working surface of these instruments is not cleaned off within 5 minutes of use, the residue will remain and cannot be removed without harm to the special surface. Also the sterilization process cannot be completed if any residue is present. If it's not clean, it cannot be sterilized.
CLEANING INSTRUMENTS
Due to daily use, and over time, all dental instruments are subject to surface changes and damage due to chemical, thermal, and/or physical impacts. Always refer to manufacturers' instructions for recommended methods and safe solutions to use during the cleaning process on instruments and equipment to ensure that instruments can be cleaned using a method available in your office. Ideally, maintaining a list of instruments to be cleaned by each method would assure uniform practices are followed by all personnel. There are basically three (3) methods of cleaning reusable dental instruments: manual cleaning, ultrasonic cleaning, and instrument washer cleaning.
All staff should be trained and understand the importance of effective cleaning and why a particular method of cleaning is preferred for one dental instrument or another. Appropriate personal protective equipment (PPE) must be worn by all employees during all phases of cleaning and preparation for sterilization. This would include utility gloves, mask, goggles, and clinic gown or jacket. Properly fitting PPE must be utilized to avoid contact with splashes, sprays, and aerosols that are present in the cleaning/sterilization area. The area for cleaning should be clearly defined to prevent cross‐contamination and maintain separation of sterile and nonsterile items. This greatly reduces the possibility of accidentally using a nonsterile item on patients. Instrument handling should be minimized, as this is where a high percentage of sharp injuries occur. Manual scrubbing is not recommended if ultrasonic cleaning or instrument washers are available.
Manual Cleaning
Always use detergents specifically formulated for manual cleaning of instruments. Never use chlorhexidine handscrub, washing‐up liquid, cleaning creams, or soap. Chlorhexidine in particular makes proteins stick to steel. Scrub instruments using long‐handled brushes with soft plastic/nylon bristles. Never use wire brushes or pot scourers as this will scratch the metal alloy instrument. On no account use wire brushes to remove rust spots. To minimize aerosol risk, do not scrub instruments under running water but rather fully submerge items to be cleaned in the detergent solution. Drying cleaned instruments properly helps prevent damage to packaging materials and reduces instrument corrosion.
Ultrasonic Cleaning
Ultrasonic units generate ultrasound waves in a liquid medium, producing tiny vacuum bubbles that allow organic debris to lift off. Ultrasonic units are ideal for cleaning the tiny crevices and openings on many dental instruments that are difficult to get to manually. Use only the manufacturers' recommended solution in ultrasonic units. Always place instruments in baskets and place the cover prior to turning on the unit. All instruments must be completely submerged or cleaning will not occur. Do not overload the ultrasonic or bind instruments together by any means because all instruments must be able to move freely within the unit for maximum cleaning efficiency. Ensure that joints or hinges are opened fully and instruments that need taking apart are fully disassembled before they are immersed in the solution for maximum cleaning.
For example, Figure 3 shows an instrument that was closed prior to cleaning and blood residue was left in the joint area, but Figure 4 shows the same instrument that was properly opened during the cleaning process in an ultrasonic unit. Ultrasonic units have been very common and popular in most dental practices because they are easy to operate, produce results up to 16 times more efficiently than manual cleaning, and remove contaminant from even the smallest cavities and crevices.
Ultrasonic insert tips may be fully immersed in a mild ultrasonic cleaning solution (pH 7.0‐10.5) or in an instrument cassette; however, avoid contact with corrosive chemicals such as chlorides, sulfates, and surface disinfectants such as phenols and iodophors because the inserts will corrode. Most manufacturers recommend a 7‐10 minute run time if instruments are free or 16‐20 minutes when the instruments are in a cassette. After cleaning either manually or in an ultrasonic cleaner, instruments must be rinsed thoroughly in purified, demineralized or distilled water to remove residual soil and detergents and then dried thoroughly. Drying cleaned instruments properly, with a non‐linting towel, will eliminate corrosion and/or microbial growth.
Testing the Ultrasonic Unit
The aluminum foil test is a simple way to evaluate and identify the general cleaning intensity and sound wave coverage throughout the ultrasonic tank for optimal performance. It is recommended that you check your ultrasonic unit on a weekly or monthly basis and keep a record of these tests. You may perform this non‐standardized test as follows:
1. Obtain any standard aluminum foil sold in a supermarket or equivalent. Cut a piece of the foil large enough to cover 1/2 to 3/4 of the tank bottom.
2. Fill tank as usual with ultrasonic cleaner, preferably a mild low sudsing solution with a neutral pH.
3. Run unit for 5‐10 minutes to properly de‐gas the solution.
4. Lower foil into the tank at an angle to avoid air getting trapped under the foil.
5. Hold or suspend the sheet of foil lengthwise across the long side of the tank and centered against the tank width. Do not allow the foil to touch the bottom of the tank.
6. Turn on the unit and allow to run for 20‐30 seconds. Turn off the unit and remove the foil for inspection.
7. Examination of the foil should reveal uniform pitting, pebbling, or perforations on the foil if the unit is working properly. Poor uniformity of the pitting, pebbling, or perforations on the foil indicate a problem and may indicate a deterioration or failure in the transducers. Repeat test before withdrawing the unit from use or sending in for repair to confirm the problem.
Instrument cassettes are gaining popularity because they increase the lifespan of instruments by keeping them from bouncing against each other during cleaning and sterilization, and keeping them in easy order for inspection. The main cause of instrument scratching is processing; instrument cassettes minimize or eliminate instrument scratching and also minimize employee handling of sharps, thus reducing injuries.
Instrument Washer/Disinfector Cleaning
Instrument washers/disinfectors are a class of device traditionally used by central sterilization services such as those in hospitals; however, more of these devices are being installed in schools and large dental practices. These FDA approved medical instrument cleaners wash, rinse, and sometimes disinfect or dry instruments, reducing the risk of pathogen transmission during subsequent instrument handling while processing. Some washers also can contain anti‐rust and anti‐corrosion chemicals to reduce damage to processed instruments. Most washers are used to clean contaminated instruments held in cassettes and must be positioned at an angle to facilitate maximum penetration of solution jetting from rotating washer arms.
Special Implant Instrument Cleaning
As a general guideline, most implant instrumentation pre‐cleaning and cleaning should follow seven basic steps:
1. Remove tissue or bone residuals by immersing the used drills in cold water. Do not use fixation agents or hot water as this will adversely affect the subsequent cleaning results. (Keep drills in wet environment until next step.)
2. Soak the drills in 0.5% enzymatic cleaning solution (with a pH level between 6 and 9) for a minimum of 5 minutes. Prosthetic implant abutments and plastic copings require special handling and must be cleaned and sterilized according to manufacturer directions explicitly.
3. Scrub the outside of the instruments with a suitable soft bristled nylon brush until all visible soil is removed. Do not use a metal brush.
4. Flush the internal channels/lumens with 20 ml cleaning solution using an irrigation needle.
5. Rinse outer side and inner sides of drills with distilled or de‐mineralized water.
6. Visually inspect for cleanliness with magnifying glass.
7. Drying is critical (minimum of 10 minutes). Additional drying can be performed manually by using compressed air.
INSPECT INSTRUMENTS FOR CLEANING EFFICIENCY AND DAMAGE
Sufficient cleaning standards are absolutely vital for successful sterilization. All instruments must be checked visually, including those processed by a washer‐disinfector. Critical areas such as handle structures, joints, or jaw serration require especially careful checking. A magnifying glass or an illuminated magnifier is an essential tool that can assure that the cleaning process has been successfully accomplished as well as find possible instrument damage such as rust corrosion, spotting, pitting, or scratching. These illuminated magnifiers are relatively inexpensive and can be found for under $15. If any debris is found it will be necessary to perform the reprocessing process again until the instruments are visibly clean. If any instruments have O‐rings, then the rubber O‐rings must be removed prior to the cleaning process and a thorough inspection of the O‐ring must be completed before re‐attaching.
Types of Corrosion
Here is how to determine if instrument discoloration is a stain or a deposit. If you believe that your instrument is stained, then perform the pencil eraser test to determine if the instrument is
truly damaged. Rub a pencil eraser over the stain. If the stain rubs off and no pitting exists then the problem is not permanent and is most likely caused by one of the following:
• Detergent residue on towels or high alkaline >8 pH detergent is being used leaving a phosphate surface deposit
• Dried blood left on the instrument
• Iodine or Betadine residue on the instrument
• Mineral deposit (usually salts or chlorine) from tap water rinse
Contact Corrosion
Contact corrosion occurs in a material combination involving stainless steel and non‐ferrous metals (German silver, brass, copper). Separate stainless steel instruments from carbon steel, anodized aluminum, titanium nitride instruments, and any carbide products during the entire process of cleaning, wrapping, or sterilization process. Stainless steel will also discolor and corrode if exposed to high concentrations of the following chemicals:
• Sodium hypochlorite (bleach)
• Aluminum or barium chloride
• Calcium chloride
• Chlorinated lime
• Ferrous chloride
• Mercury salts
• Stannous chloride
• Iodine
• Stain and tartar remover
• Bichloride of mercury
• Carbolic acid
• Citric acid
• Lysol®
• Phenol
• Hydrochloric acid
Pitting Corrosion
Pitting corrosion occurs most commonly when instruments are exposed to chlorides such as sodium hypochlorite (bleach), or stain and tartar remover. Be sure to carefully monitor the length of time the instruments are left in the ultrasonic cleaner (a maximum of 16‐20 minutes) and to thoroughly rinse and dry instruments after the cleaning process has been completed. Stainless steel instruments are especially susceptible to pitting corrosion. If pitting occurs, this leads to the creation of small holes in the metal surface and eventual weakening of the metal of the instrument.
Spotting may also appear on instruments very quickly. This is the result from the residue of minerals in tap water (such as silicon dioxide) used during rinsing and/or lack of adequately rinsed off ultrasonic cleaning solution and drying. Additionally, failure to change the ultrasonic solution on a regular basis, or failure to clean an autoclave may result in spotting. Spotting can also be caused by silicon dioxide in steam condensate from the autoclave, which is why distilled water should always be used in the steam autoclave.
Please note the spots on this hemostat; this is the corrosion from using an acid‐based cleaner or due to over‐dosing of neutralizing chemicals. Any damaged instruments such as this should be removed from use immediately. Contact the manufacturer to see if the instrument can be repaired before the corrosion escalates further.
Final rinsing of cleaned instruments in demineralized or distilled water is such an important step in instrument preservation because many instruments will discolor or stain from the minerals in normal tap water such as silicic acid (Figure 14).
Figure 15 shows a surgical scissors hinge joint with an intercrystalline crack. This is a type of corrosion of metals that attacks only the surface layers of the grains (crystallites) and penetrates objects along the grain boundaries (surfaces). The most common way to avoid the problem is to select a grade of steel that is very low in carbon, i.e., 0.03% or less, or one that is stabilized with niobium or titanium.
Wear friction corrosion is commonly seen with instruments that hinge and open and close such as scissors. This type of corrosion is very preventable with a medical grade lubricant. The photos clearly show the wearing of surface against surface. This microabrasion renders the instrument surface to become very rough, which allows further deposits or blood residue to accumulate. Regrinding and or polishing can usually repair the damage if caught early and future careful treatment with instrument oil can keep the instrument in good working order. When applying the lubricant directly to the joint area, be sure to uniformly distribute the lubricant by opening and closing the instrument repeatedly. Before autoclaving, lubricate all moving parts of instruments such as joints, box locks, ratchets, and screw joints. Water‐based lubricants should be used because they are steam penetrable. Lubricant baths are not recommended because the
solution may be contaminated from previous instruments dipped into bath. A spray or droplet lubricant is advised because they are safer, cost less, and take up less storage space.
Anti-Corrosion Measures
Anti‐corrosion measures actually begin the minute that the procedure has been completed and before the cleaning process begins. Resultant damage caused by incorrect use is not always related to the instrument material but rather how the instrument is cleaned and prepared for sterilization. The majority of instrument damage can be minimized with proper and routine instrument care.
Reviewing the Basics
• Cleaning and decontamination should occur as soon as possible after instruments and equipment are used, as dried debris can make cleaning more difficult and possibly damage the instrument surface.
• Always clean off composite material from instrument surface within 5 minutes of contact to preserve the non‐stick surface.
• Never place anodized aluminum Goldstein and Felt Instruments in an ultrasonic unit.
• Instruments should be opened and disassembled when possible before the cleaning process begins.
• An instrument exposed to high temperatures before being properly cleaned and rinsed can cause the initiation of permanent stains onto the instrument surface.
• All debris should be removed with a soft brush (never with steel brushes or abrasive items) if manual cleaning is required. Always submerge instruments in cold water to remove gross debris and help prevent coagulation of the blood present on the instrument.
• Avoid contact by way of corrosive chemicals such as chlorides, sulfates, and surface disinfectants such as phenols or iodophors with instruments.
• Following the manufacturers' instructions decreases the possibility of selecting cleaning agents that can be harmful to instruments (e.g., abrasives can damage the protective surfaces of instruments, contribute to corrosion, and impede sterilization).
• Ultrasonic cleaners should be used according to the manufacturer's operating instructions, preferably with a neutral (pH7) cleaning agent (non‐corrosive) with minimal sudsing.
• Do not batch stainless steel, aluminum, brass, or copper instruments together during the cleaning or sterilization processes because this can produce corrosion on the instrument surfaces.
• Instruments should be thoroughly rinsed after ultrasonic cleaning with distilled or de‐mineralized water because tap water contains impurities or minerals that can cause permanent stains on the instrument surface.
• Use cassettes if possible to avoid damage by contact with other instruments or damage to delicate or fine points on instruments.
• Careful drying and inspection of instruments after cleaning but before sterilizing is extremely important and allows the opportunity to identify those instruments that require additional cleaning or repair before use. Elimination of moisture helps prevent rust formation and moisture can impede the sterilization process.
• Any worn or damaged instruments should be removed from use.
• Following manufacturers' instructions, use rust inhibitors (e.g., sodium nitrate, which is effective and notably superior to other inhibitors because it is a food preservative that is relatively harmless when ingested in small quantities) on the instruments or use a dip to reduce rusting. The best approach is not to process such items through steam; instead, use dry heat or unsaturated chemical vapor sterilization. Instruments or portions of instruments and burs made of carbon steel WILL rust during steam sterilization.
• Following manufacturers' instructions, use lubricants on jointed and hinged instruments before sterilization to avoid wear friction corrosion.
• Avoid high sterilizer temperatures (above 275°F) and follow manufacturers' recommended temperature and the recommended time explicitly.
• Instruments should be stored in a clean, dry environment distant from moisture, any chemicals, or temperature fluctuations to prevent the accumulation of moisture (condensate) on instrument surfaces. Chemicals may destroy metals when in direct contact with them, or may emit corrosive vapors.
REPAIR OR REPLACE?
Many instrument manufacturers offer repair, refurbishment, re‐tipping, or sharpening of dental/surgical instruments. By all means, be sure to use a reputable instrument company when considering repairing any instrument. It is always recommended to return an instrument in need of repair back to the original manufacturer, because they know exactly what metals were used in the manufacturing process and will be able to assess if an instrument is repairable or should be discarded. Many manufacturers will not re‐tip instruments because altering the integrity of the original manufacturer's handle and tip assembly often leads to premature structural failure, resulting in serious risks to the patient and the treatment being performed. Re‐tipping is almost never recommended except for one instrument—the gold‐handled needle holder.
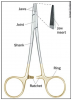
The gold rings portion of a needle holder indicate that the jaw portion contains tungsten carbide inserts. The tungsten carbide inserts are a harder metal than stainless steel and allow the practitioner to grip the suture needle more precisely with less slippage. Nongold ring needle holders are made out of the same materials (stainless steel) as the rest of the needle holder and do not have jaws that are replaceable. All gold ring needle holders' jaws will wear out, most likely at the tips of the jaws. Always inspect the jaws after use for dark colored bioburden and stains. Inspect the jaws additionally by closing the ratchet and holding the needle holder up to light. If you can see light between the jaw tips, then the tips need to be replaced. Due to the twisting motion when using needle holders, this carbide insert is susceptible to cracking. If a crack is present in the jaw, immediately take instrument out of service because broken pieces of the tungsten carbide jaw could drop into the surgical site.
Dull instruments are inefficient, lead to operator stress and fatigue, and increase the chance of traumatizing the patient. Always sharpen instruments at the first sign of dullness because the longer you wait to sharpen, the more metal will be removed and will decrease the lifespan of the instrument. If you are uncomfortable manually sharpening instruments, there are many services that will do this for you, thus allowing you to spend more time at chairside.
When replacing instruments, some manufacturers have a buy‐back program for broken or damaged instruments that will allow you a credit towards the purchase of a new instrument. Always ask if there is a buy‐back program or policy before replacing a damaged instrument.
Remember that every instrument has an expected life span, so consider negotiating an annual budget for instrument replacement. There is no doubt that instrument care and maintenance will extend the life span of every dental instrument. When purchasing quality instruments consider the old saying "you get what you pay for."
CONCLUSION
Care and maintenance of reusable instruments are crucial for successful dental treatment and instrument longevity. Well‐maintained instruments not only safeguard your patients and staff against infection, but are also essential for the outcome of the total treatment.
GLOSSARY
Alloy ‐ A mixture of metallic elements or compounds or metallic elements in varying proportions.
Anodizing ‐ A process by which an instrument (usually aluminum) is specially treated to prevent composite material from adhering to the instrument, increasing the corrosion and wear resistance and preventing discoloration of the composite or glass ionomer restoration materials. The process of creating this protective oxide coating is achieved electrolytically, which results in a metal oxide film growing on the surface of the instrument part being treated.
Batching/binding ‐ Tying together in some form or fashion instruments while submerged in ultrasonic cleaner, especially stainless steel, aluminum, brass or copper instruments.
Box‐lock ‐ The hinged portion of an instrument that allows it to open or close.
Chlorides ‐ Chemical compound containing chlorine. Most chlorides are salts that are formed either by direct union of chlorine with a metal or by reaction of hydrochloric acid with a metal. Most metal chlorides conduct electricity when fused or dissolved in water and can be decomposed by electrolysis to chlorine gas and the metal.
Corrosion - The gradual destruction or chemical attack of a surface. Usually refers to the attack of a metal surface by chemical reaction with the environment.
Electrolysis ‐ A process by which electric current is passed between two electrodes through an ionized solution effecting a chemical reaction between metals.
Enzymatic cleaner ‐ Effectively breaks down and loosens hardened debris such as dried blood, tissue and saliva.
Fretting corrosion ‐ The rapid corrosion that occurs at the interface between contacting highly loaded metal surfaces when subjected to slight vibratory motions and is due to inadequate lubrication.
Gluteraldehydes ‐ A toxic chemical that is used as a cold sterilant to disinfect and clean heat‐sensitive medical, surgical, and dental equipment.
Iodophors ‐ An antiseptic or disinfectant that combines iodine with another agent, such as a detergent.
Lumen ‐ Passageway in hollow instruments.
Phenol (carbolic acid) ‐ A form of a chemical agent used as a surface disinfectant that kills a wide range of microbes.
Pitting - A form of extremely localized corrosion that leads to the creation of small holes in the metal.
Ratchet ‐ The portion of an instrument consisting of a bar with a set of angled teeth or cogs, allowing effortless motion in one direction.
Rust ‐ The product of corrosion on iron, steel and steel alloys as a result of oxidation, a reaction with oxygen in an atmosphere containing water.
REFERENCES
Bauer, Howard. Dental Professionals Can Save Money By Reviewing Their Instrument Maintenance. Retrieved from http://www.energizingtournament.fora.pl/wazne,8/doudoune‐moncler‐gilet‐dentalprofessionals‐can‐sa,14392.html.
Bertolotti, RL and Hurst, V. "Inhibition of corrosion during autoclave sterilization of carbon steel dental instruments". Journal of the American Dental Association. 1978 Oct; 97(4):628‐32. http://jada.ada.org/cgi/content/abstract/97/4/628. Accessed July 2014.
Frontline Surgical LTD. "Instrument Care". http://www.frontlinesurgical.com/care.php. Accessed July 2014.
Hu‐Friedy website. Care and Maintenance; Composite Instruments‐Stainless Steel XTS. http://www.hu‐friedy.com/products/posterior‐xts‐composite‐instrument‐kit.html.
Integra Miltex Corporation. "Instrument care and staining information". http://www.miltex.com/prodInfo/productCare.aspx. Accessed July 2014.
Molinari, John A. "Instrument washers not dishwashers". Dental Economics Jun 2009. http://www.dentaleconomics.com/articles/print/volume‐99/issue‐6/columns/emerging‐infection‐control‐challenges/instrument‐washers‐notdishwashers.html. Accessed July 2014.
Nobel Biocare. "Cleaning and sterilization of surgical and prosthetic instruments ‐ reusable drills". https://www.nobelbiocare.com/content/dam/GMT/Produced%20Material/Sterilization%20Guideline/812/PRODMTRL_0007362_1_21120.pdf.download/76290_Cleaning%20&%20sterilization%20guidelines_GB.pdf . Accessed July 2014.
Reprocessing of Instruments to Retain Value, 10th edition. 2012. http://www.a‐k‐i.org. Accessed July 2014.
Rudolph, Matt. "How to inspect needle holders & causes of surgical instrument corrosion ‐ staining, pitting, and marking". Spectrum Surgical web site. http://www.spectrumsurgical.com. Accessed July 2014.
ABOUT THE AUTHOR
Ronda Lane is a Certified (1979) and Texas Registered Dental Assistant (2004) with 30 years of experience in the dental profession, primarily in oral and maxillofacial surgery. She is a Past President of the Texas Dental Assistants Association (TDAA) in 2004‐2005 and is currently serving as Trustee for the American Dental Assistants Association (ADAA) 2014‐17 and represents the states of Arkansas, Louisiana, Oklahoma, and Texas. She received her BS in Allied Health Education from the University of Texas Health Science Center at Dallas in 1982. Since 2006, she has taught and continues to teach the TDAA State Board of Dental Examiners approved RDA Course and Exam in Texas and worked as a chairside assistant for Dr. Chris F. Bohmfalk from 1984‐2013. Currently, she teaches dental assisting at Remington College, Fort Worth, TX. She has presented courses to her local, state, and national association. She has a deep desire for knowledge, and continues to devote herself to lifelong education in the field of dental assisting and enhancing the careers of dental assistants toward the highest standards of performance obtainable through quality continuing education. She has had several articles published in dental assisting journals including The Dental Assistant and Inside Dental Assisting, as well as The Bulletin (a TDAA quarterly newsletter).